Early noninvasive diagnosis and staging of liver cancers
Hepatocellular carcinoma (HCC) represents the most common primary hepatic malignancy and represents the second leading cause of cancer-related deaths worldwide. The incidence increased by 80% over the past 20-30 years. With a median life expectancy of up to 20 months, the outcome is generally poor despite advances in diagnosis and therapy. HCC is associated with liver cirrhosis in more than 80% of the cases. There are geographic differences due to the main risk factors. While in Asia and Africa hepatitis B infection is predominant, in Western nations it is infections with hepatitis C virus and ethyltoxic liver cirrhosis. Clinically, HCC usually remains inapparent, sometimes presenting with nonspecific signs such as pain in the right upper abdomen or unwanted weight loss. Thus, detection of HCC and other liver carcinomas remain challenging and are often too late in advanced stages. Early diagnosis would help to initiate proper diagnosis and therapy with curative intent. Therefore, our group focuses on non-invasive diagnosis of liver cancer and liver dysfunction to improve patient care.
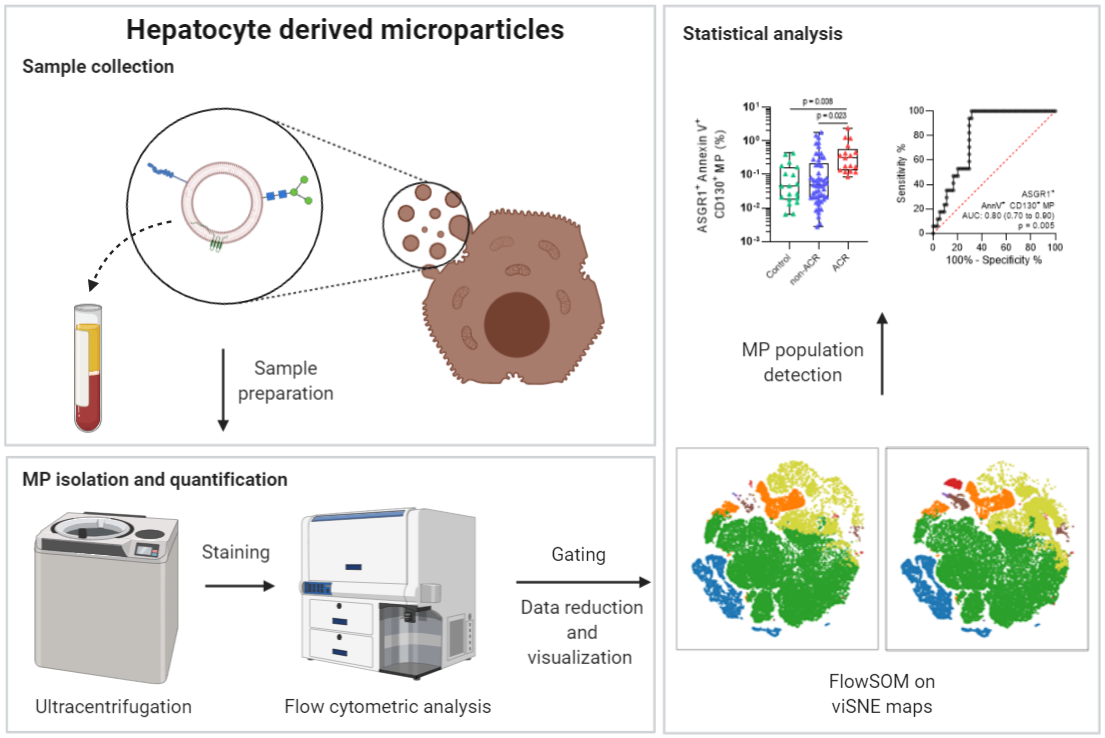
The concept of liquid biopsy can be described as non-invasive alternative to the classic biopsy, in which information about the condition of an organ can be derived from a blood sample and the molecules circulating in it (Figure 1). We have recently explored the importance of liver-specific extracellular particle population. Liver dysfunction and acute cellular rejection subsequent liver transplantation is diagnosed by liver biopsy and concomitant histological analysis, representing the gold standard in clinical practice. Yet, liver biopsies are invasive, costly, time intensive and require expert knowledge. We are able to diagnose acute cellular rejection non-invasively. A novel extracellular particle populations in samples were identified using visualization of t-distributed stochastic neighbor embedding (viSNE) maps and self-organizing maps (FlowSOM) algorithms. It was found that the ASGR1+ CD130+ annexin V+ particles exhibited the highest accuracy for predicting acute cellular rejection (Figure 2). In addition to the analysis of extracellular particles, our group is engaged in metabolomics analyses and analyses of the phenotypic properties of immune cells to open up new diagnostic applications (Figure 3).
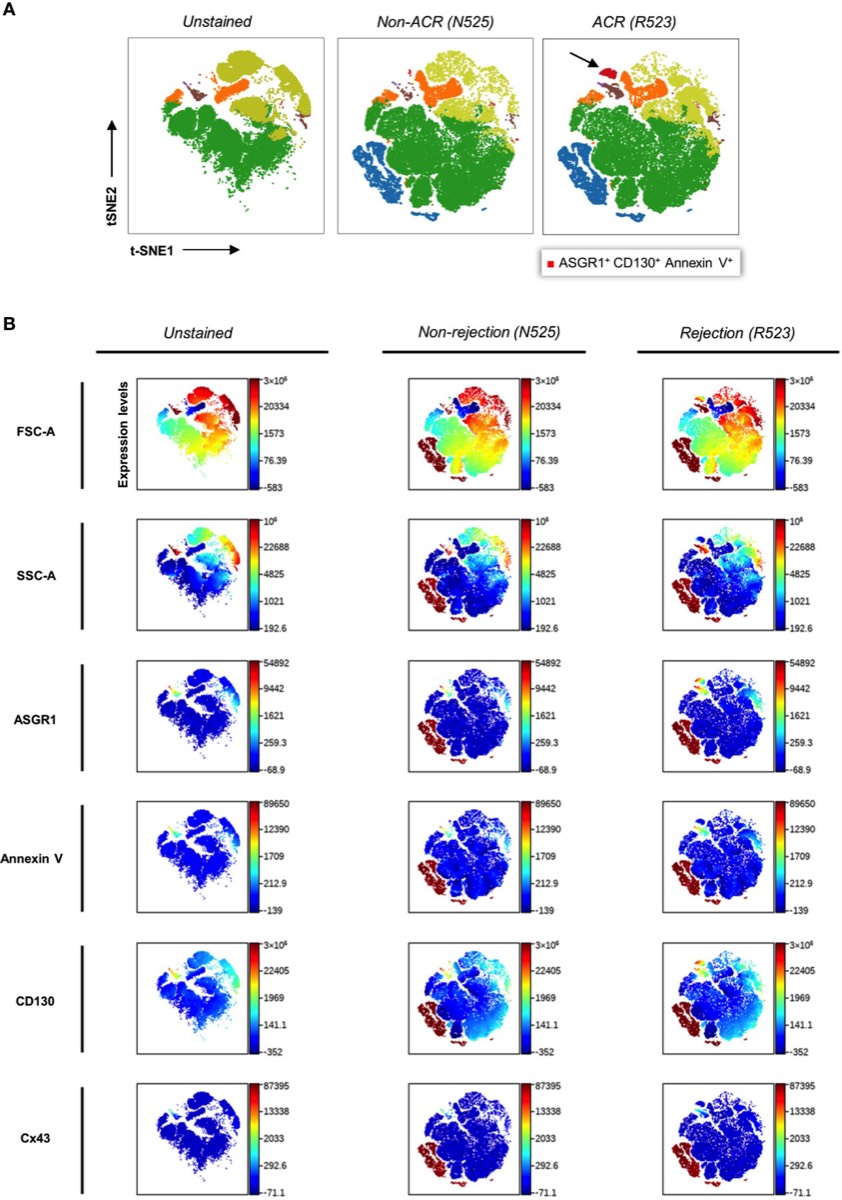
Figure 2: Clustering and color coding with FlowSOM and viSNE for identifying EP subpopulations via density plots. t-SNE (t-distributed stochastic neighbor embedding), an unsupervised nonlinear dimensionality reduction algorithm, was used to fit six-dimensional data into two dimensions. All clusters were created via FlowSOM analysis. The arrow indicates the novel ASGR1+CD130+AnnV+ EP population. (Kaan et al. 2021)
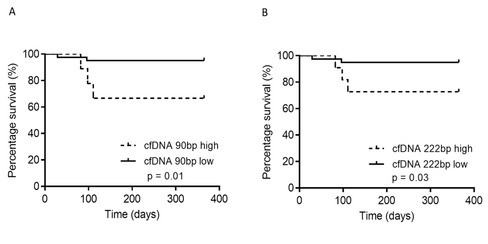
Figure 3: cfDNA analysed according to survivors and non-survivors (1-year follow-up). Patients with cfDNAlow displayed a superior 1-year survival rate; the 1-year survival rate with cfDNAlow (90 bp) was 95%, compared with 66% for cfDNAhigh (p = 0.01). Mann–Whitney U test; Log-rank test (for survival); * p < 0.05; ** p < 0.01. (Krenzien et al. 2020).
Project-related publications:
Sensing Acute Cellular Rejection in Liver Transplant Patients Using Liver-Derived Extracellular Particles: A Prospective, Observational Study
K Kamali et al., Frontiers in Immunology 12, 1601
Increased cell-free DNA plasma concentration following liver transplantation is linked to portal hepatitis and inferior survival
F Krenzien, et al., Journal of clinical medicine 9 (5), 1543
Diagnostic biomarkers to diagnose acute allograft rejection after liver transplantation: Systematic review and meta-analysis of diagnostic accuracy studies
F Krenzien, et al., Frontiers in immunology 10, 758
Correlation of cell-free DNA plasma concentration with severity of non-alcoholic fatty liver disease
T Karlas, et al., Journal of translational medicine 15 (1), 1-9


