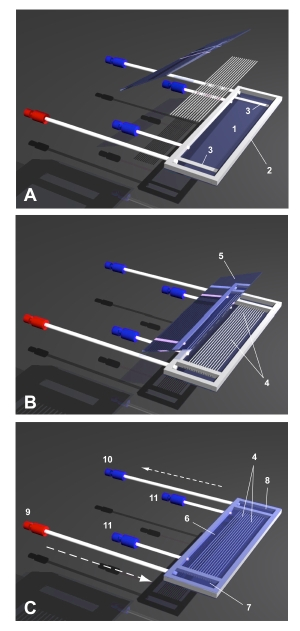
SlideReactor
The SlideReactor is a simple hollow fiber based bioreactor construction suitable for light microscopy and time-lapse video observation. The SlideReactor offers a cell compartment separated from a medium inflow and outflow compartment. Cell compartment access ports enable easy filling of the cell compartment with cell suspension, as well as fixation of the tissue. For more complex procedures or full access to all the cells, the bioreactor can be opened easily by cutting the silicone seal with a scalpel. Due to its simple design and the utilization of standard materials, it could serve as a suitable, cost-efficient tool to evaluate the behavior of cells cultured between hollow fiber capillaries.
SlideReactor
The first experiments showed the feasibility of this concept:The SlideReactor’s cell compartment is completely visible and – due to the use of optical quality glass – ideal for video time-lapse microscopic observation of the behaviour of different cell types cultured between hollow fiber capillaries. Cell divisions as well as movement and growth of the cells can be recorded. The morphology of the cells as well as the growth of the tissue was comparable to the controls. The cells were viable, actin filaments and nuclei showed no abnormalities compared to the control cultures.
The SlideReactor may serve as a simple tool to evaluate the cell-to-cell and cell-to-hollow fiber interaction. The device enables the characterization of cell behavior under controlled conditions and the analysis of the influence of medium supplements on the cell viability and tissue integrity. It may be useful for the comparison of different types of hollow fibers, e.g. for future use in bioreactor-based extracorporeal liver assist devices.
For evaluation the SlideReactor was charged with primary human liver cells obtained after partial liver resection and different cell lines (HuH7, C3A and WiDr). During cell culture the cells are analyzed by time-lapse video microscopy. After termination of the culture and a first evaluation by phase contrast microscopy the cells are fixed with PFA, stained with fluorescence dyes (phalloidin, CK18 and DAPI) and analyzed via immunofluorescence microscopy. As control, cells were cultured in standard monolayer techniques.
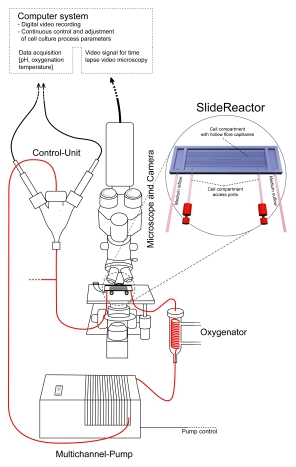
Schematic experimental set-up: To ensure a continuous observation of the cultures, the SlideReactor is positioned on a microscope, continuously perfused by a roller pump. Probe-heads within the medium reservoir allow continuous determination of relevant process parameters. The perfusion system the microscope and the camera are placed in a heating unit adjusted to 37°C.
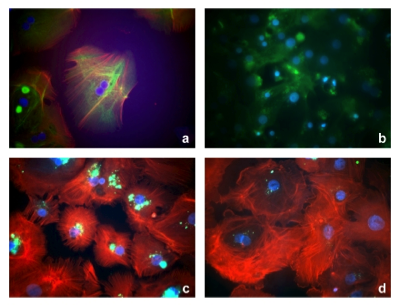
Proof of Concept

Cells cultured in the SlideReactor and the respective controls: Primary human hepatocytes (a,b), WiDr cells (c,d), primary human fibroblasts (e,f) and primary human keratinocytes (g,h). Magnification: 100x (a-d) and 200x (e-h).
All cell types evaluated in this study can easily be cultured, observed and analyzed inside the SlideReactor. The perfusion set-up allows the regulation of all relevant process parameters serving the individual demands of the cultured cell type. The culture period varied according to the cell types: the primary human cells with no or low proliferation rate were cultured for seven days; whereas the culture of the highly proliferating cell lines was terminated after approximately five days.
The cell morphology and function of primary human hepatocytes was analyzed: after cell isolation and purification, the cell suspension had a viability of 87.7 ± 3.6% (mean ± standard deviation, n=5 isolation procedures). The primary human hepatocytes were relatively large in size (about 30 µm in diameter) and showed normal morphological appearance: they had the typical polygonal shape, slightly granular cytoplasm with vesicular inclusions and one or two nuclei. No abnormalities concerning their adherence and spreading characteristics were observed when cultured inside the SlideReactor. No structural deviations were detected when analyzed via phase contrast microscopy. One million seeded cells were sufficient to cover the entire surface.
Albumin and CK18 were analyzed as protein expression parameters, while actin and CK19 were used as positive and negative controls, respectively. Primary human hepatocytes were positive for albumin. The staining of CK18 showed clearly structured intermediate filaments located throughout the entire cytoplasm. However, the protein was most frequently detected around the nucleus. The actin filaments were straight and located throughout all adhesion areas, especially at the contact areas between the cells.
Staining of the cells with the apoptosis-indicating fluorescent dyes CytoDeath and TUNEL-kit did exclude apoptotic processes in the majority of the cells. As a positive control, primary human hepatocytes isolated from the same specimen were exposed to irradiation on the third day of culture (100Gy). After two more days, these cells were detaching, smaller in size and the cytoskeleton was unstructured; subsequently, apoptosis in these cells was verified by staining.
The editors of Tissue Engineering have chosen one of the figures showing fluorsecent staining of primary human hepatocytes cultured within the SlideReactor as cover-art!

